the same sample spectra collected at different time by mid-infrared spectrometer is different

Submitted by xiaowan on 22 August 2013 - 9:19am
Forums:
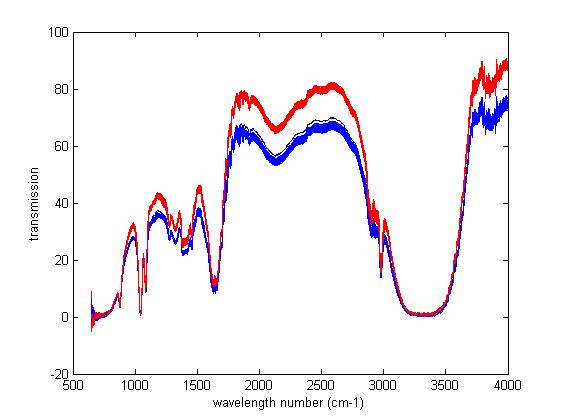
There are two kinds of liquid substances, which is the same exactly, with a mid-infrared spectrometer measured one per day. The experimental conditions remained the same, and both of the background are null samples. However, the two spectra have been inconsistent, and there is a difference of 20 with the transmission as the ordinate. The detail sees in figure with the difference between blue and red.
Uploaded Files:
Uploaded Images:
hlmark
22 Aug 2013
Xiaowan - there is information related to your question in the nature of the differences between the two spectra. The main detail of importance is that the two spectra are spectrally similar, but have an offset between them. Furthermore, the offset appears to be roughly proportional to the transmittance reading. This implies that the cause of the difference is a physical effect wherein more light is passing through the sample optics (and sample) for one spectrum than for the other. One likely possibility is movement of some part of the optics between collecting the two spectra. One likely culprit is the sample cell. Other things you need to look at are any parts of the instrument that normally move either during or between spectra. They may not be returning to their proper home position. Alternatively, they may not be returning to the same position during the scan. Examples could include any mirrors, optical stops, physical stops, apertures or any adjustable component that might affect the optical path.
\o/
/_\
Permalink
td
22 Aug 2013
Hi Xiawan and Howard,
Was the same cell used for all the readings or did you use different cells for your different liquids? The small variations within the same sample my be due to small temperature varaitions.
Howard and I have both assumed that these measurements were made using an NIR spectrometer and the scale is wavelength (nm).
Best wishes,
Tony
Permalink
hlmark
22 Aug 2013
Tony - it actually doesn't matter what units the wavelength scale represents, since there don't seem to be any noticable wavelength shifts, although careful reading of the plot reveals that the X-scale is explicitely labelled as cm-1. In any case, the largest changes are all at the top of the plot, which would become baseline shifts if the plot were converted to absorbance. What's not so clear from the transmission spectrum is whether the absorbance readngs at all wavelengths would be shifted by the same amount, although I rather suspect that they would. If Xiowan could convert his spectra to absorbance and post those spectra, it would clear up this question for us, at least.
Even though that type of change could be corrected mathematically, Xiaowen is quite right to be concerned about this lack of reproducibility of his raw data. Since he is dealing with liquids and not solids, that simply should not happen, and the presence of it is a symptom of some (currently unknown) phenomenon affecting his data. It may be something as simple as non-reproducibiliy of the cell positioning, or something more complex, that might require a service call from the manufacturer to diagnose and repair.
\o/
/_\
Permalink
ianm
23 Aug 2013
Determine the spectral precision and also the repeatability of the instrument itself. Take 8–10 scans of the same sample, with thorough re-blending of the sample, and re-loading the sample each time. Leave the 8th (or 10th) scan in the sample cell, and re-scan it 7–9 more times without touching the sample or sample cell between scans. Calculate the standard deviation (SD) of the raw absorbance data from the first series of scans at at least two different wavelengths, followed by the coefficient of variance (CV). This will give the reproducibility of the scans with re-loading. Then determine the SD and CV of the absorbance data at the same wavelengths for the last 8–10 scans that will have been taken without touching the sample cell. This will give the repeatability of the instrument, and should be much lower than the SD/CV of the reproducibility.
If the reproducibility is very much higher than the repeatability it is the sample presentation that is the main cause of the problem. If the instrument repeatability is poor that is the time to consider that the instrument itself may be responsible for the difference in absorbance from day to day.
Posted on behalf of Phil Williams
Permalink
td
23 Aug 2013
Hi Phil,
These are liquid samples so I do not think blending is a problem.
I had assumed that they were NIR spectra but now I believe that they are mid-IR . Xiawan said he was using a mid-IR spectrometer and the scale if nm or cm-1 is in the IR region.
Xiawan,
I think your spectra are mid-IR but this is an NIR forum! But now we have started I'm sure we would like to help you.
Please could you answer some questions:
1) What is the make and model of your spectrometer?
2) Please can you tell us the identity of your samples or if not something about them?
3) Please give complete experimental details for obtaining the spectra.
Howard,
I think it does matter if these are NIR or MIR spectra. I think they are MIR and it is more than 30 years since I was seriously looking at MIR spectra!
I still think the variation between red and blue spectra could be sample thickness.
Best wishes to all,
Tony
Permalink
hlmark
23 Aug 2013
If we take Xiaowan's spectrum at face value, then the range 500-4000 cm-1 corresponds to 20-2.5 microns, or 20,000 to 2,500 nm. In either set of units, then, it's definitely a mid-IR spectrum. The large, broad troughs at ~2,200 cm-1 and ~3,300 cm-1 look like -OH (or possibly even water) in the mid-IR. Considering the presence of sharp bands at ~2,900 cm-1 and elsewhere in the spectrum, indicative of -CHn, my guess is that this sample is an alcohol.
But I think the problem Xiaowan is asking about is independent of the wavelength range and even the sample, and has to do strictly with the absorbance measurement, and how it's being affected. As Xiaowan said, since he's working with a liquid, optical scattering is not an issue, and neither is "segregation" of the mixture.
Besides being labelled "Transmission", the Y-scale goes from 0 to 100, which is far beyond any rational value for absorbance, but perfectly normal for transmittance. Since it's a transmission measurement, the "baseline" is at rougly 80-90%T, and that is what is varying. If cell pathlength were the problem, we would expect the "baseline" to be constant, and the changes in absorbance to be most evident at the spectral absorbance peaks, which show up as spectral minima (troughs) in the transmission domain.
The variations we see on the spectrum are consistent with an opaque object intruding into the sample channel beam, thus cutting off a constant fraction of the light at all wavelengths, and the wavelengths of higher energy (i.e., higher transmittance) would show a larger offset than those at lower energy; and this is what we observe. If this hypothesis is correct, it would result the same absorbance change at all wavelengths. That's why I asked Xiaowen to compute and plot the two spectra in absorbance units.
Other effects that mimic an opaque object intruding into the sample beam would cause the same result. One possibility that occurs to me, for example, is movement of the cell so that an edge of the beam is cut off (vignetted). Movement of internal optics in the instrument, that would cause similar changes to the optical path, would also have the same result.
Some of the questions brought up about the hardware are very pertinent. For exmaple, is the instrument single-beam or double-beam? Etc. Knowledge of the manufacturer and model could allow us to look up this information. If Xiaowen wants us to be able to help him, however, he's got to be more forthcoming with the information he releases to us. I get an impression that he's holding back, and telling us only the minimum amount he can.
\o/
/_\
Permalink
hlmark
23 Aug 2013
It just occured ot me that another possibility would be a change in the stray light.
\o/
/_\
Permalink
xiaowan
12 Sep 2013
There are two bottles of liquor with the same batch from the winery. Samples were scanned with a NEXUS 670 FT-IR spectrophotometer (Nicollet, USA) in the reflectance mode. The groove side containing liquid is the crystal ZnSe 45°. Before scanning, the liquor spectrum was acquired to substract from a background scanned in an empty cell. Then, we removed a certain amount of liquor (called a sample), which is appropriate with not overflowing, and poured into the groove. The spectrum of each sample was the average of 16 successive scans. 50 times were operated continuously. Such, there are 50 spectrum collected in this bottle.
The next day, reboot machine and re-scan the background and collect 50 spectrum of the other liquor with the same operation. Moreover, all devices have not been moved in the laboratory.
The transmission plot is the figure 1, and the figure 2 is the absorbance data. All blue lines are the first liquor with 50 spectrum, the same to red lines.
Permalink
jjakhm
12 Sep 2013
Xiaowan,
What type of reflectance measurements did you perform, e.g. specular, diffuse, internal, etc.? Can you provide the identity of the accessory (There are many for the instrument you listed)? Do you have the internal polystyrene standard wheel and the Val-Q functionality available (I believe these options are not included in the base cost of the instrument… this would help to identify if any instrument related issues are present, along with the other diagnostic functions that are included with the base instrument and Omnic software…)? Did you use Omnic? I ask because the images are not something that Omnic would produce…
I would like to provide some feedback regarding the sample preparation and measurement phases of analysis since the potential instrument related issues seem to be well covered in the previous replies. I also have experience with an instrument very similar to the one that you are using.
For example, if the measurements were done in attenuated total reflectance (ATR) mode, inclusion of air bubbles on the internal reflection element may be something to consider. Maybe think about how you treated the samples prior to measurement and how they were dispensed onto the IRE. In the past I have observed air entrainment and bubbles, in liquid samples, adhered to the surface of the IRE on a Golden Gate accessory (Nexus 470 FTIR). This would produce results consistent with Howard’s mention of the intrusion of an opaque object into the beam path. This situation becomes more likely given the assumption that I made regarding the way you performed the measurements (The way you described the process is a little unclear to me):
You used the phrase “50 times operated continuously,” I took that to imply that you dispensed 1 aliquot of liquid onto the “groove” and measured the same aliquot 50 times (since you mentioned nothing about cleaning, re-dispensing, etc.).
Had the spectra represented measurements each of a different aliquot, I would suspect an instrument related issue. But if these represent 100 spectra from two aliquots, it is very possible (and perhaps the simplest explanation) that something was present in your sample (red spectra) that persisted throughout the measurement process. Once a bubble adheres to the surface of the IRE it is not likely to let go.
Reflectance mid IR measurement absorbance intensity (as it relates to depth of penetration) is also susceptible to changes in sample refractive index. For example, a refractive index change of ~0.03, given a typical ATR-FTIR set-up, could result in an ~5% difference in depth of penetration, and therefore roughly 5% difference in absorbance intensity over the spectrum (effective path-length = # reflections x depth of penetration), which is about the level that occured between your two data sets. However, I’m not sure how likely this is given your measurement environment (temperature fluctuations etc.) and sample storage conditions (contamination, evaporation of mixture components, etc.). I suppose it is not out of the question that a confluence of conditions could occur and conspire to influence the refreactive index to that degree.
Hope that my reply is not too late and helps in some way.
Best Regards,
Jason
Permalink
hlmark
12 Sep 2013
Jason brought up a good point: bubbles. Whether ATR or standard transmission cell, bubbles could cause the typr of shift observed.
Xiaowan should redo the experiment, taking care to ensure that no bubbles can form. Without knowing more about the setup, we can't be more specific as to how to sccomplish that, but even microbubbles, of the sort that form when water is left standing for a long time and air bubbles form on the container could create that sort of effect.
\o/
/_\
Permalink
ahmed_nrc
21 Mar 2014
Hi
I think there is a problem in the device itself. I think this devices need to not be switched off as long as it continues working it gives a good reading. as well as, the most important to differentiate between the samples is not the looking of the peaks but is the chemometric analysis and the validation
Ahmed
Dr. Ahmed A. Afifi
Permalink