[in-line monitoring of mammalian cells culture] Scattering issue caused by high cell densities

Submitted by david_didion on 9 June 2014 - 11:47am
Dear people of the imp forum,
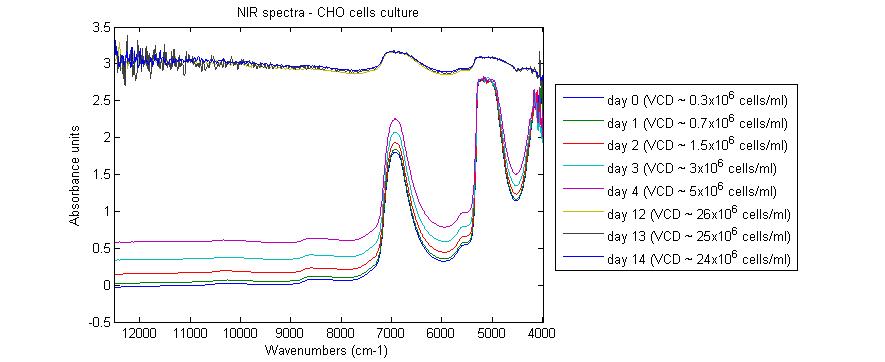
I try to develop in-line predictive models for monitoring nutrients and metabolites concentrations in suspension high cell density cultures of CHO cells. I use a Bruker Matrix-F FT spectrometer and a Hellma transflectance immersion probe with an optical path length of 1 mm. The problem I have to deal with is the increase of the log(1/T) values throughout the bioprocess (see attached image). Spectra from day 5 to 11 are not available because of undesirable interruption of spectra acquisition. On day 12 (and doubtless well before), log(1/T) values are so important that I am afraid spectra are of no use.
To my understanding both scattering effect caused by cells and fouling of the analysis slit of the probe due to viscous culture broth, both increasing with the cell density, might contribute to these high log(1/T) values. It must be noted that fouling of the probe is just a supposition because of vigorous agitation (280 rpm). To reduce the scattering effect it is tempting to decrease the optical path lentgh but it is not possible because of material limitation. Moreover decreasing the optical path length would lead doubtless to probe fouling. Pratically I have doubt I can further optimize the log(1/T) values with the available optical path lenghts (2 mm and 5 mm) and I don't see any other measurement parameter to test for solving this problem. What would you suggest me?
Do you think that diffuse reflectance or transmission probe might be more appropriate to this application?
Thank you in advance!
David, PhD student at the University of Mons in Belgium
Additional measurement parameters: resolution = 16 cm-1, 64 coadded scans/spectrum, background collected in air in the bioreactor, length of optical fibers = 2 m, temperature of the bioprocess = constant (36.8 °C)
Sarah Bellini
10 Jun 2014
Hello David,
I an currently working on my PhD thesis about spectral measurement in dense microalgal culture media. I have thouroughly investigated light interactions with optically complex media, that is why your problematic rings some bells.
What I call "optically complex media" here are media that appear heterogeneous from the light point of view, meaning they are composed of elements that are bigger or of the same order of magnitude as the incident wavelength. I imagine that your cells size are about some microns, what makes that your culture medium is indeed turbid, and all the more turbid that the cell density in the medium is high.
Photons incident on a medium can have different behaviour: they can be absorbed, scattered or regularly transmitted (meaning that they cross directly throughout the medium without any interaction with matter). In fact, absorption and scattering originate from the same physical effect between the incident electric field of light and the discrete electric charges (electrons and protons) composing the matter. The discrete electric charges of matter are set into oscillatory motion by the incident electric field of light, and they de-energize either by re-radiating the gained energy as light (scattering) or by transforming it into another form of energy, generally heat (absorption). My point here is to illustrate that for any media, scattering and absorption phenomena always occur: what we call in real life absorbing media simply present absorption properties much higher than scattering ones, but they also scatter light anyway.
In my sense, you observe such high absorbance values because your culture medium is highly concentrate, what increase both absorption and scattering phenomena. Stastically, in a crowd of cells, an incident photon has the best chance to interact with matter, being either absorbed or scattered in a direction different from the original one. At high cell densities, you may even encounter multiple scattering, which means that a photon will encounter successive interaction phenomena with matter (being for example scattered once, finds another cell on its way and being finally absorbed). The result is that very little photons will finally manage to cross throughout your dense medium, what explains the low transmittance values and the high absorbance. Reducing the pathlength is indeed a way to measure more transmitted photons at the other side of your sample (it is actually a way to reduce the cell density). You could even think about diluting the cell culture, and integrate it in your modelling. But I think that you may obtain more interesting results in changing your measurement confirguration, meaning that you could try to measure light in other directions than only the regular one (basic configuration of transmittance spectrophotometers).
You could for example measure transmitted light in a greater solid angle by using an integrating sphere setup adapted to your spectrophotometer. I bet that you would measure more light, and obtain more exploitable spectra. As you said you could also measure the diffuse relfected light by using also an integrating sphere. The best would be to combine all the measurements you can do, which would surely makes your modelling more robust. Indeed, your cell media have optical properties that are probably not isotropic. Moreover depending on the cell density and the scattering regime, their geometrical properties may vary. I strongly advise to make at least three measurements: regular transmittance (just as you do, with the smallest pathlength), diffuse transmittance and diffuse reflectance. Make sure that you do all the measurements in the same conditions (same quartz cell in particular). And integrate them all in your modelling.
I hope my answer was not too long and could give you some ideas to solve your problem.
Good luck!
Sarah, PhD at IRSTEA, Montpellier
Sarah Bellini
PhD student - Optical Engineer
IRSTEA, France
Permalink
david_didion
10 Jun 2014
Hello Sarah,
Thank you for your quick, clear and generous answer!
Indeed the size diameter of the CHO cells is about 15 µm.
I do not use a transmittance probe but well a transflectance (or transflection) probe, which as you surely knows, collects both transmitted and reflected parts of the incident beam. For information, the reason why we choosed this type of probe is summarized by Arnold et al. (Use of at-line and in-situ near-infrared spectroscopy to monitor biomass in an industrial fed-batch Escherichia coli process, Biotechnology and bioengineering, 2002): "(...) The mode of data acquisition depends on the optical properties of the broth. Generallly, for low cell density bioprocesses, transmission is used, whereas for strongly light-scattering fermentations reflectance is used. (...)". We choose the transflectance because of the broadly evolutive conditions in a high density mammalian cell culture.
I agree with your analysis and will do my best to get new probes with integrating spheres.
Thank you!
David
Permalink